 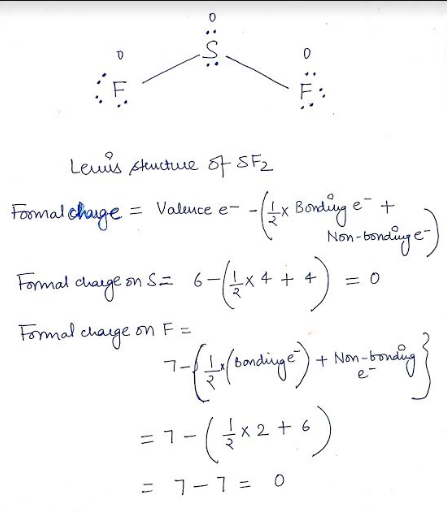   
The straw Lewis structures for each of the following molecules for a H two is a little structure h each for be hbr. Lewis Structure of SF2 Lewis Structure is nothing but an arrangement of valence electrons between different atoms. Sulfur is single-bonded to each of two fluorine atoms and has two lone pairs as well. In the SF 2 is Lewis structure Sulfur S which is the least electronegative and goes at the center of the Lewis structure. For the SF2 Lewis structure we first count the valence electrons for the SF2 molecule. Can you rematch with someone you recently unmatched on Tinder. SO 4 2- N 2 O XeO 3.Ģ0 e-F prefers 1 bond and follows the octet rule. A step-by-step explanation of how to draw the SF2 Lewis Structure. Given that the central sulfur has 4 electron pairs surrounding it 2 bonding and 2 non-bonding VESPER predicts that these are arranged in a tetrahedron to a first approx.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
